AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Carbon tetrachloride formula lewis structure10/12/2023 
When reading about smog production or ozone depletion, consider the bond strengths of the chemicals involved in this environmental problems. This method is tolerant to esters and carbonyl compounds because it does. Dichlorocarbene can also be obtained by dechlorination of carbon tetrachloride with magnesium with ultrasound chemistry. 
Explanation: It name of following compound, dicarbon tetrahydride, state its chemical. Carbon Tetrachloride: Symbol, Structure, Uses, Formula Spletcarbon tetrachloride. Although this chemical species has not been isolated, it is a common. Carbon tetrachloride is a compound having the molecular formula CCl4. Carbon Tetrachloride AMERICAN ELEMENTS Lewis Dot Structure and Polarity of. For example, the triple bond found in carbon monoxide is harder to break than one of the double bonds found in carbon dioxide. Dichlorocarbene is the reactive intermediate with chemical formula CCl 2. For this reason, it is more difficult to break or cleave a multiple bond than it would be a single bond or a bond of a lower order (when comparing the same connected atoms). Multiple bonds involve more that one type of connection. 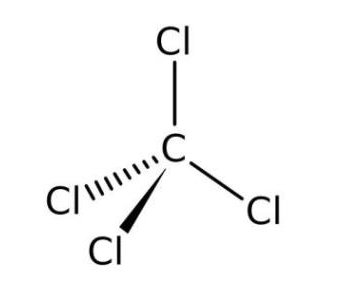
The carbon atom has two lone pairs and two bonding pairs. The formula of the carbonate ion is CO 3 2. The carbonate ion (see figure below) consists of one carbon atom and three oxygen atoms and carries an overall charge of 2. Together, they comprise a single ion with a 1+ charge and a formula of NH 4 +. If an octet can be achieved with a single bond, do not incorporate multiple bonds into the structure. In the Lewis structure, the carbon atom (C) is in the center, bonded to two hydrogen atoms (H) and two bromine atoms (Br).Each bromine atom has three lone pairs of electrons and one bonding pair. The ammonium ion (see figure below) consists of one nitrogen atom and four hydrogen atoms. N2H4 Lewis In the Lewis structure of CI4 structure there are a total of 32 valence. Let's look at a few molecules that utilize multiple bonds:Ītoms that could employ multiple bonds would be oxygen, nitrogen, sulfur, phosphorus, and carbon. Carbon tetrachloride is a compound having the molecular formula CCl4. Although it is mathematically feasible, halogens will not use multiple bonds to become stable either. Please note the rules above on which atoms will never achieve octets (hydrogen and boron). If a single bond does not work, then you may incorporate a multiple bond. In covalent bonding, using double (four total electrons) or triple (six total electrons) is an option in order to achieve a stable octet.
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
